Metallic Characteristics
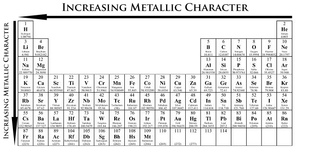
Metallic Characteristics are the chemical properties of metals. This set of properties can be given the name 'Metallic Character' so that it can be measured among all elements.
Metallic Characteristics include:
- Ability to be reduced
- forming chlorides through ionic bonding
- forming basic oxides
- Ability to displace Hydrogen from diluted acids
Metallic Character increases from right to left on a period, and increases down a group. Therefore Helium has little or no Metallic Character while Francium expresses many Metallic Characteristics.
Metallic Characteristics include:
- Ability to be reduced
- forming chlorides through ionic bonding
- forming basic oxides
- Ability to displace Hydrogen from diluted acids
Metallic Character increases from right to left on a period, and increases down a group. Therefore Helium has little or no Metallic Character while Francium expresses many Metallic Characteristics.
