Ionic Size
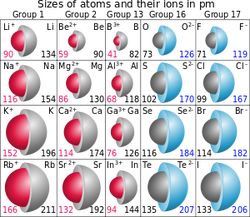
Atoms become ions when they gain or lose electrons. When gaining electrons the atom becomes negatively charged and is called an anion. If it loses electrons it becomes positively charged and is called a cation.
Anions are larger than their 'parent atoms' because they have gained electrons but not protons, meaning it is still the same element but it now has a charge. On the other hand, Cations are smaller than their parent atoms because they now have less electrons.
Ionic size is a measurement of the size of an atoms relative ion. Like atomic size it is measured from the centre of the nucleus to the outermost occupied electron shell.
Anions are larger than their 'parent atoms' because they have gained electrons but not protons, meaning it is still the same element but it now has a charge. On the other hand, Cations are smaller than their parent atoms because they now have less electrons.
Ionic size is a measurement of the size of an atoms relative ion. Like atomic size it is measured from the centre of the nucleus to the outermost occupied electron shell.
