Electronegativity
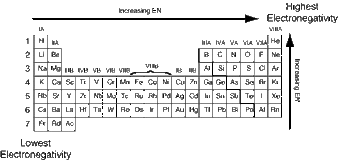
Electronegativity is a measure of an atom's ability to attract electrons to itself. It is affected mainly by two properties; the atom's atomic number and how far it's valence electrons sit from the nucleus.
The higher the electronegativity, the more the element, or even compound, attracts electrons toward itself. Interestingly, electronegativity cannot be directly measured, and must be derived from calculations involving other atomic or molecular properties.
Electronegativity increases across a period, and decreases down a group, so the highest electronegativities are found in the upper right hand corner of the periodic table, and the lowest in the bottom left.
The higher the electronegativity, the more the element, or even compound, attracts electrons toward itself. Interestingly, electronegativity cannot be directly measured, and must be derived from calculations involving other atomic or molecular properties.
Electronegativity increases across a period, and decreases down a group, so the highest electronegativities are found in the upper right hand corner of the periodic table, and the lowest in the bottom left.
