Introduction
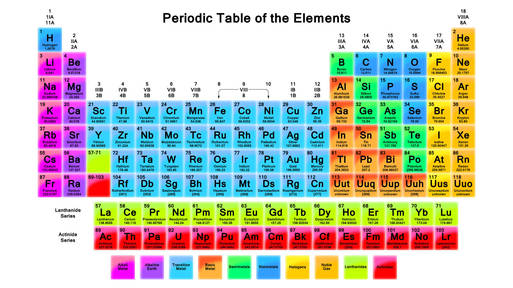
Welcome to periodictrends! This website is a class project which will go into detail regarding the many trends of elements on the periodic table. Any students that need a reference feel free to use the site :)
Why the Periodic Table is arranged the way it is
There are several reasons that the periodic table is pieced together in a very specific and consistent manner. Primarily, as you move along the Periodic Table, each atom has one more proton than it's predecessor, and one less than it's successor, in their basic states. Because of the way that atoms work and function, this also sets the trends in the table, which are what this site is here to discuss in further detail.
Groups and Periods
Groups are the vertical columns of the Periodic Table. There are 18 groups. Elements in a group share similar characteristics among their outermost shell, and have the same number of valence electrons. Valence Electrons are the electrons in the outermost occupied shell of an atom.
Periods are the horizontal rows of the Periodic Table. There are 7 periods. Elements in a period have the same number of occupied electron shells. These are the electron shells that contain orbiting electrons.
Groups and Periods are both ways of categorising the elements, and work in conjunction with each other to logically order the periodic table. However, they both express different things, and can be shown separately, but the table as a whole cannot work without both.
Periods are the horizontal rows of the Periodic Table. There are 7 periods. Elements in a period have the same number of occupied electron shells. These are the electron shells that contain orbiting electrons.
Groups and Periods are both ways of categorising the elements, and work in conjunction with each other to logically order the periodic table. However, they both express different things, and can be shown separately, but the table as a whole cannot work without both.
