First Ionisation Energy
First ionisation energy is the minimum amount of energy required to remove the highest energy electron from an atom. This force is measured in kj/mol (kiloujoules per mole).
Joules are a measurement of energy equal to the amount of work or energy used to apply 1 Newton of force through 1 metre.
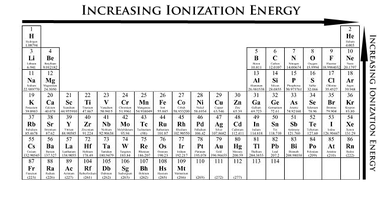
First ionisation energy increases across a period, and decreases down a group, as shown in the diagram (left). This means the Helium has one of, if not the highest, First ionisation energy compared to all elements in their standard states.
Joules are a measurement of energy equal to the amount of work or energy used to apply 1 Newton of force through 1 metre.
First ionisation energy increases across a period, and decreases down a group, as shown in the diagram (left). This means the Helium has one of, if not the highest, First ionisation energy compared to all elements in their standard states.
